Ozone Depletion
Ozone Depletion
What is Ozone depletion
At any given time, ozone molecules are constantly formed and destroyed in the stratosphere. The total amount, however, remains relatively stable. The dynamic equilibrium between creating and breaking down ozone molecules depends on temperature, pressure, energetic conditions and molecule concentrations. The concentration of the ozone layer can be thought of as a stream's depth at a particular location. Although, water is constantly flowing in and out, the depth remains constant.
The concentrations of ozone vary naturally with sunspots, seasons, and latitudes. The processes of variation of ozone are well understood and predictable. Scientists have established records for several decades which show distribution of ozone during the natural cycles. It has been observed that each natural reduction in ozone levels has been followed by a recovery cycle. However, convincing scientific evidences have shown that the ozone layer is being depleted well beyond changes due to natural processes.
Ozone depletion occurs when the natural balance between the production and destruction of stratospheric ozone is shifted towards destruction. The equilibrium can be disturbed for instance by other molecules which react with the ozone molecules, and thereby destroying them. If this destruction process is fast and the creation of new molecules is too slow to replace the destroyed ozone molecules, the equilibrium will get out of balance. As a result, the concentration of ozone molecules will be reduced. An upset in this balance can have serious consequences for life on Earth, and scientists are finding evidence of the changed balance. As a result, the concentration of ozone within the protective ozone shield is decreasing.
When very stable man-made chemicals containing chlorine and bromine enter into the atmosphere, and reach the stratosphere, these chemicals are broken down by the high energy solar UV radiation and release extremely reactive chlorine and bromine atoms. These undergo a complex series of catalytic reactions leading to destruction of ozone.
Large fires, certain types of marine life and volcanic eruptions also produce chlorine molecules. These are chemically active but most of it gets converted into water soluble inorganic compounds which gets washed down by rain and only traces reach the stratosphere. However, United States Environment Protection Agency (USEPA) exper iments have shown that Chlorofluorocarbons (CFCs) and other widely used chemicals produce roughly 85% of the chlorine in the stratosphere.
It was also believed that large volcanic eruptions can have an indirect effect on ozone levels. Although, Mt. Pinatubo's 1991 volcanic eruption did not increase stratospheric chlorine concentrations, it did produce large amount of tiny particles called aerosols. These aerosols increase chlorine's effectiveness in destroying ozone.
The aerosols only increase depletion because of the presence of CFC- based chlorine. In effect, the aerosols increase the efficiency of the CFC siphon, lowering ozone levels even more than that would have otherwise occurred. Unlike longterm ozone depletion, this effect is however short-lived. The aerosols from Mt. Pinatubo have already disappeared, but satellite, ground based, and balloon data still show ozone depletion occurring closer to the historic trend.
Chlorofluorocarbons (CFCs)
For over fifty years, CFCs were thought of as miracle substances. CFCs were first created in 1928 as non-toxic, non-flammable refrigerants, and were first produced commercially in the 1930's by DuPont. The first Chlorofluorocarbon was CFC-12, a single carbon with two chlorines and two Fluorines attached to it.
These have been used in many ways since they were first synthesized in 1928. They are stable, inert, non-flammable, low in toxicity, and inexpensive to produce. Over the period, CFCshave been used as aerosols, refrigerants, solvents and foam blowing agents in other smaller applications. Other chlorine and bromine-containing compounds include methyl chloroform, a solvent, Carbontetrachloride (CTC), an industrial chemical, halons, extremely effective fire extinguishing agents, Hydrochlorofluorocarbons (HCFCs) mainly used as a refrigerant and foam blowing agent and methyl bromide, an effective fumigant used in agriculture and grain storage.
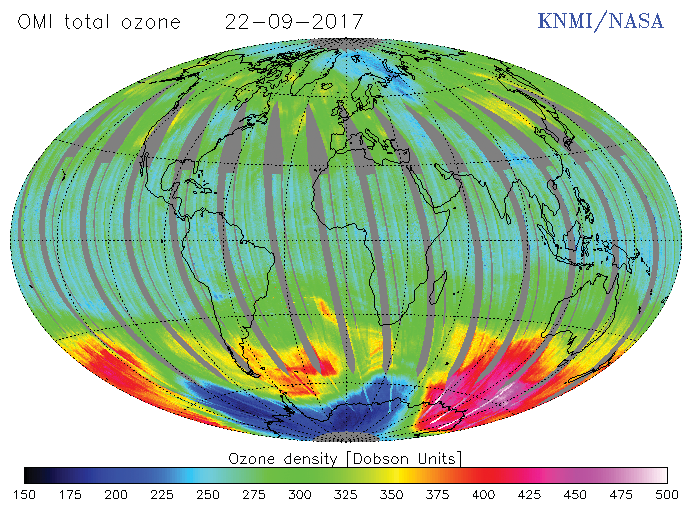 All of these compounds have very long atmospheric life which allow them to be transported by winds into the stratosphere. During the past 5 decades, Ozone Depleting Substances (ODSs) including CFCs have been emitted into the atmosphere in large quantity which has resulted in depletion of the ozone layer resulting increased UV-B radiations on the Earth surface.
All of these compounds have very long atmospheric life which allow them to be transported by winds into the stratosphere. During the past 5 decades, Ozone Depleting Substances (ODSs) including CFCs have been emitted into the atmosphere in large quantity which has resulted in depletion of the ozone layer resulting increased UV-B radiations on the Earth surface.
The largest losses of stratospheric ozone occur regularly over the Antarctica every spring, leading to substantial increase in UV levels over the region. A similar, though weaker, effect has been observed over the Arctic. There was enough evidence that ozone levels decrease by several percent in the spring and summer in both hemispheres at middle and high latitudes.
There is also fall in ozone levels during the winter at these latitudes in the southern hemisphere. The higher levels of loss of ozone have been noticed since late 1970s.
In the early 1970s, researchers began to investigate the effect of various chemicals on the ozone layer, particularly CFCs, which contain chlorine. They also examined the potential impacts of other chlorine sources like chlorine from chlorination of water, industrial plants, sea salt and volcanoes etc. The chlorine released from such applications and from other sources readily combines with water and other chemicals and form compounds which do not reach the stratosphere. In contrast, CFCs are very stable and do not dissolve in rain. Thus, there are no natural processes that remove the CFCs from the lower atmosphere. Over a period of time, the CFCs diffuse into the stratosphere where these interact with UV rays of short wave length which breaks them.
The CFCs are so stable that only exposure to strong UV radiation breaks them. When that happens, the CFC molecule releases atomic chlorine. It has been estimated that one chlorine atom can destroy over 100,000 ozone molecules. The net effect is the destruction of ozone, faster than its natural creation. To return to the analogy comparing ozone levels to a stream's depth, CFCs act as a siphon, removing water faster than normal and reducing the depth of the stream.
Last Modified : 7/31/2023
This topic provides information about overview of ...
This topic provides information about Earth's Atmo...
Climate Change, Effects, Insect Pests, Population ...
This topic provides information about Climate chan...
