Advisory to start rapid antibody based blood test for COVID-19
Advisory to start rapid antibody based blood test for COVID-19
Overall testing for COVID-19 using real-time RT PCR is being used in the country for COVID 19 testing. At the same time, it is planned to deploy Rapid Test kits (blood based) for use in response to COVID-19 situation.
ln this regard, National Task Force deliberated with experts for ascertaining use ofthese rapid test kits. Draft of the suggested algorithm was also discussed in detail with technical experts from the Ministry of Health and Family Welfare and inputs were included in the final version which is feasible for implementation in the field conditions.
Advisory to start rapid antibody based blood test for COVID-19, in clusters (with containment zones), and in large migration gatherings/evacuees centres is now made available.
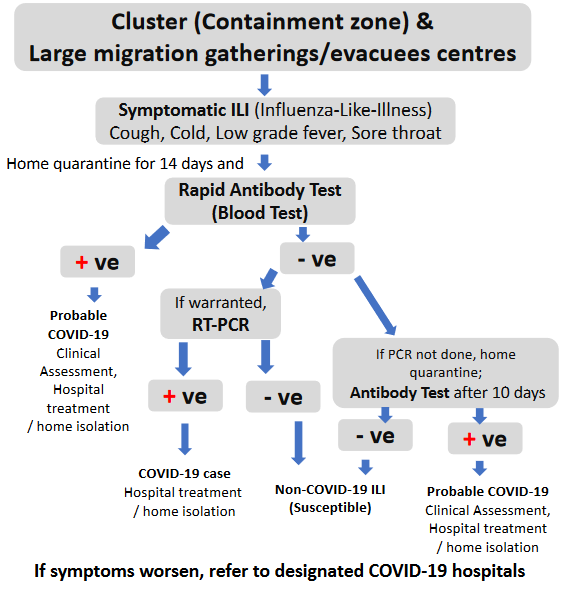
Strategy for areas reporting clusters (containment zone) and in large migration gatherings/evacuees centres
Cases of Influenza Like Illness (ILI) to be monitored in health facilities. Any surge in cases to be monitored and brought to the notice of Surveillance Officer/CMO for additional investigation.
As a matter of abundant precautions, all symptomatic ILI persons should be advised home quarantine for 14 days.
At facility level, symptomatic ILI individuals to be tested using rapid antibody tests.
Antibody test negative:
- If warranted, confirm by real-time RT-PCR using throat/nasal swab.
- RT-PCR negative: Likely non-COVID-19 ILI
- RT-PCR positive: Confirmed COVID-19 Case and action as per protocol to be initiated for isolation, treatment and contact tracing.
OR
- If real-time RT-PCR not done, home quarantine and repeat antibody testing after 10 days of the last rapid antibody test.
- Antibody test negative:Likely non-COVID-19 ILI.
- Antibody test positive:there is possibility of recent infection, quarantine for another 10 days.
Antibody test positive: After clinical assessment,treatment in hospital or isolationas per protocol. Action as per protocol to be initiated for contact tracing.
If symptoms worsen, refer to designated COVID-19 hospitals.When home quarantine is not feasible, consider facility-based quarantine.
General Guidelines
- Healthcare workers doing the rapid antibody test to use gloves, mask, and head covers.
- Healthcare workers collecting throat/nasal swab to follow standard national infection control guidelines.
- The rapid antibody tests approved by US-FDA/CE-IVD or non-CE-IVD validated by ICMR-NIV with marketing approval by DCGI be used.
- In order to ensure that all such cases are monitored and necessary action is initiated with respect to infectious disease management, details of all test results shall be uploaded in ICMR portal.
- All such organizations are duty bound to register themselves to ICMR portal and upload the data in real-time.
- Failure to do so, they will be held liable to action under Disaster Management Act, 2005.
Strategy for use of rapid test
Source : Ministry of Health and Family Welfare
Last Modified : 5/3/2021
Provides information about the Standard Treatment ...
This topic contains the information related to Nat...
Ministry of Education has issued guidelines regard...
National Directives for COVID-19 Management
